Effective use of Firmagon (degarelix) for advanced prostate cancer

Firmagon (Degarelix) is a competitive, reversible GnRH receptor blocker, for patients with advanced hormone-dependent prostate cancer. Where Injection technique is critical; Nora Sullivan, a specialist nurse at Hibernian Healthcare, explains that very slow injection (over two or even two and a half minutes) is the key to minimising the risk of injection site reactions.
by Christine Clark: Prostate cancer is now the most common cancer in Europe for men and the third most common cancer overall. Some 417,000 new cases were diagnosed in 2012(CRUK1) and there were just over 92,000 deaths. (CRUK 2a) In the UK, about 30 men die from prostate cancer every day and the majority of deaths are in the over-75 age group. (CRUK 2b) However, survival has improved considerably. Forty years ago, more than two in 10 men diagnosed with prostate cancer survived their disease for at least ten years, now it is more than eight in 10. Much of this increase is attributed to earlier and wider detection of prostate cancer as a result of the use of the tumour marker, prostate specific antigen (PSA). (Ref CRUK 3) Prostate cancer is more common in older men, those with a family history of prostate cancer and in black African and Afro-Caribbean men. (CRUK 5)
Advanced prostate cancer describes locally invasive disease (tumour stage T3 or T4) or metastatic disease. Locally invasive disease can cause obstructive uropathies and one of the problems of spread of disease to bones can be spinal cord compression. The American Joint Committee on Cancer (AJCC) TNM system is used to determine the stage of the disease. It is based on five pieces of information – the extent of the tumour (T), whether it has spread to lymph nodes (N), the presence of metastastis (M), the PSA level at diagnosis and the Gleason score (based on prostate biopsy findings). (Ref American Cancer Society)
Androgen deprivation therapy (ADT) is one of the key elements of treatment for advanced prostate cancer. The rationale for this is that testosterone is essential for the growth and proliferation of prostate tumours. ADT results in regression of androgen-dependent tumours. For many years ADT has relied on either orchiectomy or treatment with long-acting gonadotrophin releasing-hormone (GnRH) analogues. These measures still form part of modern treatment pathways (Ref NICE, European Association of Urology guidelines) but are not without problems. Orchiectomy results in a rapid fall in testosterone levels but can be associated sexual dysfunction and negative psychological effects. GnRH agonists cause an initial surge in luteinising hormone (LH) and follicle stimulating hormone (FSH) levels and a corresponding increase in the level of testosterone. Chronic exposure to a GnRH agonist results in down-regulation of GnRH receptors and androgen suppression. The initial surge can be associated with tumour growth and a ‘symptomatic flare’ that can involve worsening bone pain, urinary obstruction, spinal cord compression and fatal cardiovascular events as a result of hypercoagulability. (Ref Drudge-Coates p87a) The clinical effects of a flare may be prevented by pre-treatment with an antiandrogen. (Ref Drudge-Coates p87b) The recently-introduced, direct-acting GnRH blocker, degarelix, which does not cause an initial surge, may offer some advantages.
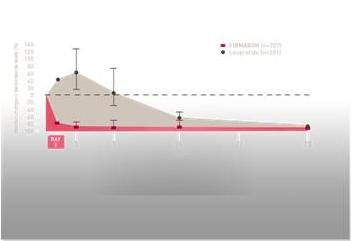
Degarelix is a competitive, reversible GnRH receptor blocker. It reduces the release of LH and FSH and brings about a rapid suppression of testosterone to castrate levels (<0.5ng/ml) without provoking a surge or flare of symptoms. (Ferring SPC, sect 4.2) A 12-month, randomized, comparative study investigated the efficacy and safety of two different degarelix monthly dosing regimens with a starting dose of 240 mg (40 mg/ml) followed by monthly subcutaneous doses of 160 mg (40 mg/ml) or 80 mg (20 mg/ml), in comparison to monthly intramuscular administration of 7.5 mg leuprorelin in patients with prostate cancer requiring androgen deprivation therapy. (Ferring SPC, sect 5.1) The results showed that testosterone levels fell to castrate levels in 96% of patients in three days whilst in the leuprorelin group there was an initial rise in testosterone levels followed by a steady fall over the next 10 days.
PSA levels fell with both treatments, but more rapidly with degarelix. The authors concluded that degarelix was non-inferior to leuprolide (leuprorelin) at maintaining low testosterone levels over a one-year treatment period. (Klotz 2008). These results were subsequently confirmed by a pooled analysis of studies comparing degarelix with GnRH agonists in which individual patient data from 1920 patients were analysed. (Klotz 2014 p1102). The results showed that there was a significant improvement in PSA progression-free survival (PSA PFS) and overall survival (OS), as well as reduced incidence of joint, musculoskeletal, and urinary tract adverse events, compared with GnRH agonists. (Klotz 2014 p1101) A further study – an extension of the original comparative study – examined the safety and efficacy of degarelix over a five-year period. (Crawford 2014) The results showed that the significant PSA-PFS benefit established for degarelix over leuprolide during the 12-month study remained consistent at five years. (Crawford 2014 p1122) The most common treatment-related adverse events were injection-site reactions. These included pain, erythema, swelling, and nodules. Reactions were most common after the first injection (31% of 276 initiation doses) and occurred less frequently with subsequent doses (2.5% of 9142 maintenance doses). (Crawford 2014 p1124a) In total, 2% of patients discontinued degarelix treatment because of injection-site reactions. (Crawford 2014 p1124b)
The frequency of injection site reactions in the published studies calls attention to the importance of good injection technique. Firmagon (degarelix) injection has been designed as a depot subcutaneous injection and is therefore administered differently from short-acting subcutaneous injections. In order to work effectively the drug needs to be reconstituted correctly and administered very slowly by deep, subcutaneous injection. The drug has to be reconstituted, immediately before administration, using the diluent provided. This forms a gel that acts as a depot in the subcutaneous tissue. The concentration of the drug in the gel is critical for correct performance and the packs (for both initial and maintenance doses) have been designed to ensure that the correct concentrations can be made up easily. The initial dose pack contains two vials of drug powder containing 120mg and two preloaded syringes each containing 3 ml of solvent. The maintenance dose contains one vial of drug powder containing 80mg and one preloaded syringe containing 4 ml of solvent. Vial adapters are also included in each pack to ensure that the vial is pierced correctly and the reconstitution operation is needle-free.
It is important to dissolve all the drug powder so that a clear fluid containing no undissolved particles or solids is administered. The diluent should be injected slowly into the vial containing the powder. It is important to avoid shaking the drug to prevent foam formation but it may be swirled very gently to facilitate dissolution. If powder adheres to the vial above the liquid surface, the vial can be tilted gently. A ring of small bubbles is acceptable but foam is not as it makes it difficult to draw up the dose. A dose of 3 ml is then withdrawn into the syringe, a needle is fitted (included in the pack) and the dose is administered by deep subcutaneous injection (see below). The dose must not go into a vein or into muscle. The site of the needle should be checked before depressing the plunger by aspirating gently to check for blood. If blood appears in the syringe, the product must be discarded and a new dose made up.
The injection is given in the vicinity of the navel, but avoiding areas that will be subject to pressure such as the belt or waistband area or near the ribs. Other areas, such as the back of the arm, that are used for other types of subcutaneous injections are not suitable for Firmagon because there is insufficient subcutaneous tissue. For the initial dose, two separate injections are given in different sites.
Injection technique
The injection technique is critical as poor technique can result in considerable pain and discomfort for the patient. Nora Sullivan, a specialist nurse at Hibernian Healthcare, explains that very slow injection is the key to minimising the risk of injection site reactions. Over the past seven years Ms Sullivan has worked with about 100 patients receiving both first doses and maintenance doses of Firmagon. Site reactions such as pain and itching, in both situations, can be reduced if the injection is given over two or even two and a half minutes, she explains. Typically, a slow injection might take 30 seconds, but Firmagon should be given much more slowly, she advises. Patients who have had previous injections given by their GP or practice nurse have frequently commented that her technique causes little or no site reaction. “A steady, very, very slow push” is required, she emphasises.
Two other points are important for correct administration: the skin should be gently but firmly pinched between thumb and forefinger to elevate the subcutaneous tissue and then the needle should be inserted about halfway down the slope at an angle of not less than 45°and the injection is administered. In this way the Firmagon injection is placed deep in the subcutaneous tissue. The drug will be steadily released from here over the next four weeks.
Ms Sullivan acknowledges that it can be difficult to convey the importance of using an appropriate injection technique to nurses and other clinicians “because it goes against the grain to take this much time to administer an injection”. The key things to remember are not to shake the vial when reconstituting the drug and to administer it very slowly. “This is one area where we can make a difference to the patient experience – a positive injection experience really helps”, says Ms Sullivan.
Finally, it is important to discuss the process with the patient to help to shape his expectations. He may have looked at advice on websites of patient support groups or read descriptions of bad experiences in internet discussion forums. It is wise to warn the patients that injection site reactions (ISRs) are not uncommon with the first dose but are usually much milder for subsequent doses. The reactions usually settle after a few days and do not interfere with the action of the drug. Discomfort can also be managed using simple analgesics (paracetamol or ibuprofen) and/or cool packs.
For many men with advanced prostate cancer, ADT is an important component of treatment. Degarelix offers rapid reduction of testosterone levels without the risks of an initial surge and remains effective and tolerable over a long period. The concept of a depot subcutaneous injection needs to be clearly understood. The use of a very slow injection technique can improve the patient experience considerably and it is something that should be emphasised to GPs and practice nurses. In addition, patients should be warned about the possibility of injections site reactions, especially with the first dose and advised to use analgesics or cool packs to relieve discomfort.
References
CRUK 1
http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer/incidence#heading-Four accessed June 2015
CRUK 2
http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer#heading-One accessed June 2015
CRUK 3
http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer#heading-Two accessed June 2015
CRUK 4
http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/prostate-cancer/risk-factors#heading-Three
CRUK 5
American Cancer Society. The AJCC TNM staging system http://www.cancer.org/cancer/prostatecancer/detailedguide/prostate-cancer-staging
NICE clinical guideline 175. Prostate cancer: diagnosis and treatment. January 2014 https://www.nice.org.uk/guidance/cg175/resources/guidance-prostate-cancer-diagnosis-and-treatment-pdf
European Association of Urology Guidelines on Prostate Cancer 2015 http://uroweb.org/guideline/prostate-cancer/
Drudge-Coates L. GnRH blockers: a changing paradigm in the management of prostate cancer. Urological Nursing 2009;3: 85-92
Ferring SPC
https://www.medicines.org.uk/emc/medicine/21701/SPC/Firmagon+120mg+Injection/
Klotz L, Boccon-Gibod L, Shore ND Andreou C, Persson BE, Cantor P, Jensen JK, Olesen TK, Schröder FH. The efficacy and safety of degarelix: a 12-month, comparative, randomized, open-label, parallel-group phase III study in patients with prostate cancer. BJU Int. 2008 Dec;102:1531-8.
Klotz L, Miller K, Crawford D, Shore ND, Tombal B, Karup C, Malmberg A, Persson BE. Disease Control Outcomes from Analysis of Pooled Individual Patient Data from Five Comparative Randomised Clinical Trials of Degarelix Versus Luteinising Hormone-releasing Hormone Agonists. European Urology 2 0 1 4; 66: 1 1 0 1 – 1 1 0 8
Crawford D, Shore ND, Moul JW, Tombal B, Schröder FH, Miller K, Boccon-Gibod L, Malmberg A, Olesen TK, Persson BE, Klotz L. Long-term Tolerability and Efficacy of Degarelix: 5-Year Results From a Phase III Extension Trial With a 1-Arm Crossover From Leuprolide to Degarelix. UROLOGY 83: 1122e1128, 2014