Metoject subcutaneous injection pen (methotrexate) pen – type autoinjector approved in Japan – Eisai
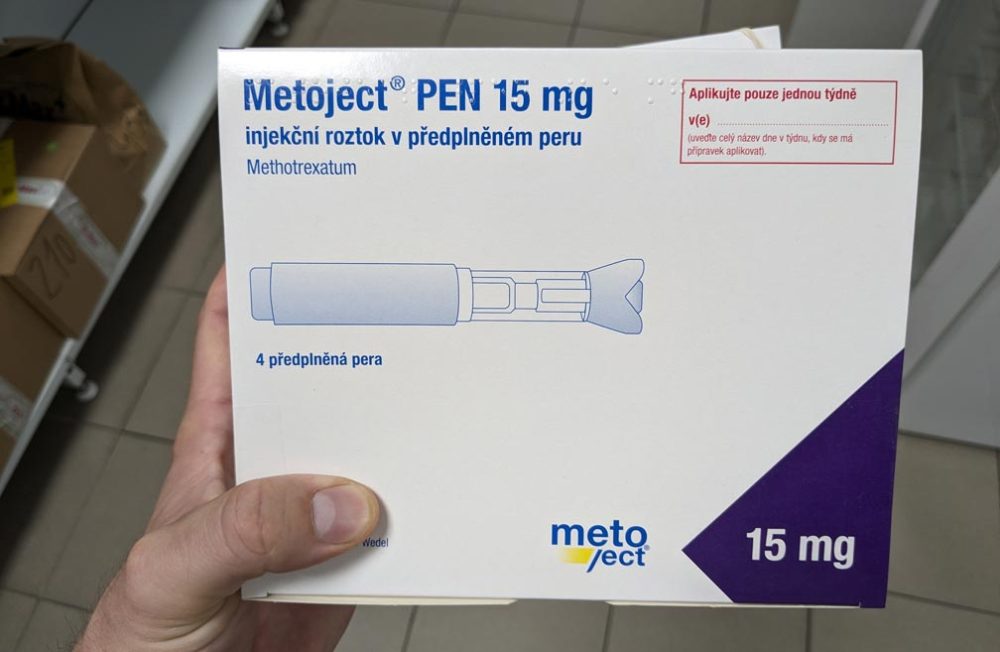
Eisai Co., Ltd. and nippon medac Co., Ltd., a subsidiary of medac Gesellschaft für klinische Spezialpräparate mbH announced that they have obtained manufacturing and marketing approval from the Japanese Ministry of Health, Labour and Welfare (MHLW) for the additional formulation of Metoject Subcutaneous Injection for the anti-rheumatic agent, pen-type autoinjector “Metoject Subcutaneous Injection 7.5mg Pen 0.15mL, 10mg Pen 0.20mL, 12.5mg Pen 0.25mL and 15mg Pen 0.30mL” (methotrexate).
Based on the license agreement signed by Eisai and medac GmbH in May 2019, nippon medac will hold the marketing authorization of Metoject, while Eisai will be responsible for product distribution of Metoject in Japan.
This formulation incorporates the previously MHLW-approved Metoject Subcutaneous Injection pre-filled syringe formulation in a pen-type autoinjector and was developed to reduce the burden on patients and improve safety during self-injection. The drug can be self-injected in two steps (1- removing the cap, 2- pressing the pen against the skin). The built-in needle cover prevents the needle from being seen prior to administration and automatically locks after administration to prevent accidental skin puncture.