EU drug watchdog warns of fake Ozempic pens
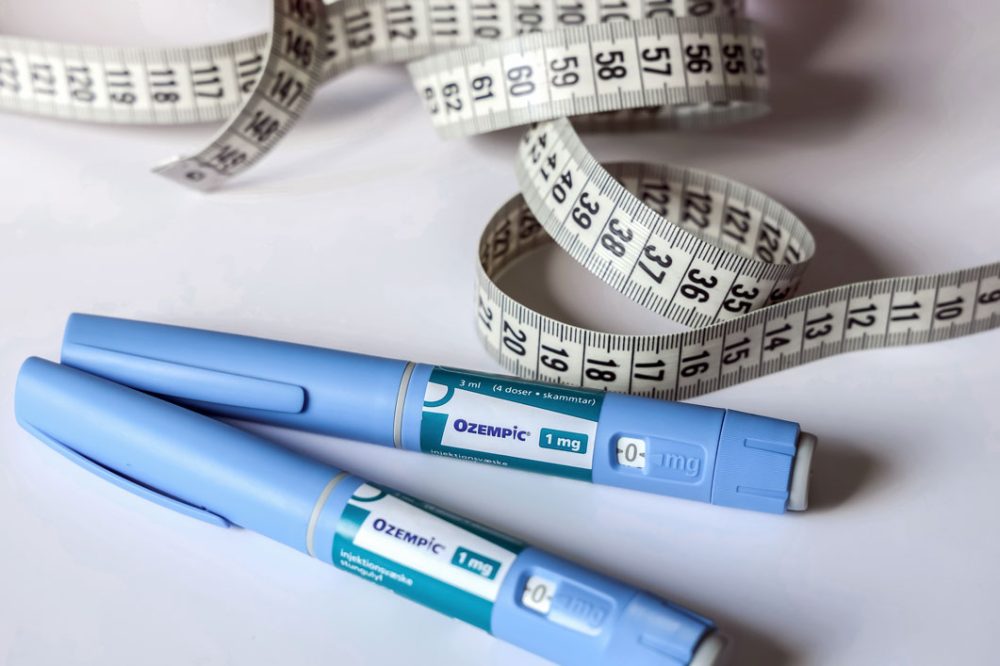
The European Medicines Agency (EMA) is warning patients and healthcare professionals to beware of pre-filled pens falsely labelled as Ozempic, a diabetes medicine linked to weight loss.
National authorities notified the EU regulator after the pens, which are labelled in German and appear to have originated from wholesalers in Austria and Germany, were found in several locations in the EU and the UK.
The pens have batch numbers, 2D barcodes and unique serial numbers from genuine Ozempic packs. In the EU, each medicine pack has a unique 2D barcode and serial number so that it can be tracked in an EU-wide electronic system. When the packs of the falsified Ozempic were scanned, the serial numbers were shown to be inactive, thereby alerting operators to a potential falsification.
There are differences in the appearance between the falsified pen and the original pen. There is no evidence that any falsified pens have been dispensed to patients from legal pharmacies and there are no reports of harm to patients in relation to the falsified medicine, the EMA said in a statement.
Ozempic pens (semaglutide, 1 mg, solution for injection) are in high demand across Europe and globally. The drug helps to lower blood sugar in people with diabetes by helping the pancreas to make more insulin. There have been shortages of the medication in several countries, with some social media influencers highlighting its off-label use in weight loss. While it is not authorised for this purpose, it is associated with modest reduction in patients’ weight.
The EMA is assisting national authorities in their investigations. Wholesalers and pharmacies in the impacted countries have been warned about the suspicious offers of Ozempic to wholesalers. In addition, parallel distributors across the EU have been alerted.
In the meantime, the German and Austrian regulatory authorities have issued statements of non-compliance with good distribution practices (GDP) to the concerned wholesalers in their countries for not following required procedures, including compliance with security measures.





