UK MHRA grants marketing authorisation for Cibinqo for adults and adolescents with moderate to severe atopic dermatitis – Pfizer
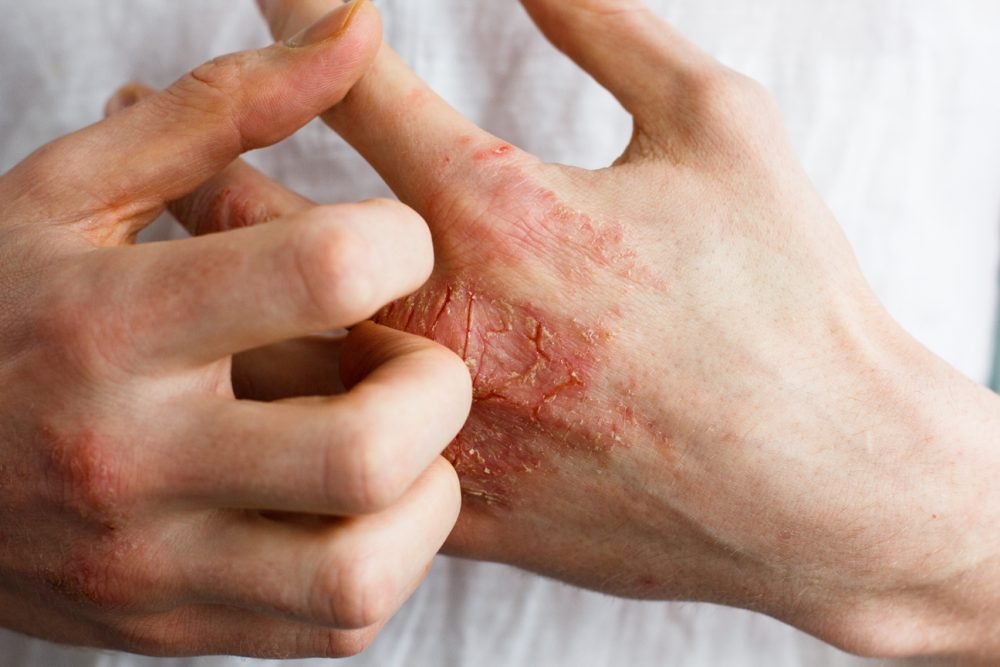
Pfizer Inc. announced that the UK Medicines and Healthcare products Regulatory Agency (MHRA) has granted Great Britain marketing authorization for Cibinqo (abrocitinib), an oral, once-daily, Janus kinase 1 (JAK1) inhibitor, for the treatment of moderate to severe atopic dermatitis (AD) in adults and adolescents aged 12 years and over, who are candidates for systemic therapy.
Abrocitinib is licensed in Great Britain in recommended doses of 100mg and 200mg. This is the first marketing authorization worldwide for this treatment.
Regulatory applications for abrocitinib have been submitted to countries around the world for review, including the United States, Australia, Japan, and the European Union..





